Restoring immune balance by targeting pathogenic T cells
Rosnilimab is a first-in-class antibody that selectively depletes pathogenic T cells in the periphery and inflamed tissues — aiming to restore immune homeostasis without broad immune suppression.
A pathogenic T-cell depleter
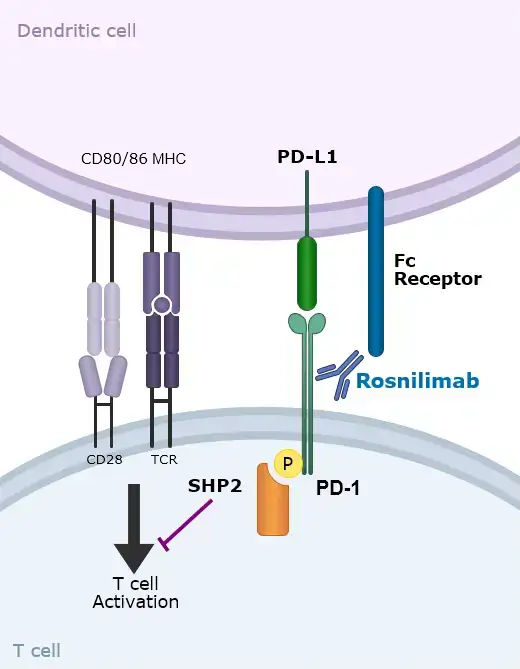
Rosnilimab is an antibody engineered to selectively deplete pathogenic T cells in both the periphery and inflamed tissues.
- By reducing these disease‑driving cells while sparing naïve T cells, rosnilimab aims to restore immune homeostasis without broadly suppressing the immune system
- Through this targeted approach, rosnilimab reduces T‑cell activity, limits B‑cell recruitment and decreases plasma‑cell–mediated autoantibody production, addressing multiple drivers of autoimmune pathology
Rosnilimab is currently being studied in rheumatoid arthritis (RA).
RA: A chronic, systemic autoimmune disease
RA develops when the immune system mistakenly attacks the body’s own tissues. Joint pain, swelling and stiffness are hallmark symptoms, but RA can also affect the skin, eyes, lungs, heart and blood vessels.
Persistent inflammation drives progressive joint damage and may significantly impair daily life.
Why rosnilimab is a promising approach
Rosnilimab has demonstrated a potential best in disease profile in RA, with a compelling safety and tolerability profile and JAK like efficacy through six months that remains durable for at least three months off drug.
Highlights from the Phase 2b clinical trial
Well tolerated, with:
- Rapid clinical and symptomatic improvement across all patient groups by three months — including in difficult to treat populations
- Continued improvement through six months
- No treatment-related serious adverse events
- No malignancies across ~185 patient-years of exposure
These results support rosnilimab’s potential to deliver meaningful, durable benefit while maintaining a favorable safety profile.
Scientific presentations & publications
Access the latest data, abstracts and peer-reviewed literature about Rosnilimab